Immune competence largely depends on immune cell renewal and clonal expansion of T- and B-cell populations. When the immune system ages, the adaptive immunity deteriorates because of progressive declines of naïve T and B cells and decrease of absolute numbers of T and B lymphocytes[1]. How to determine the aging status of the immune system? And what are the possible tailor-made actions to rejuvenate immune system based on the genetic make-up? Now, there is a simple way to assess the aging of the immune system testing the Telomere Length. This test can also reveal the changes of telomere length caused by the intervention regime you prescribed to your patients.
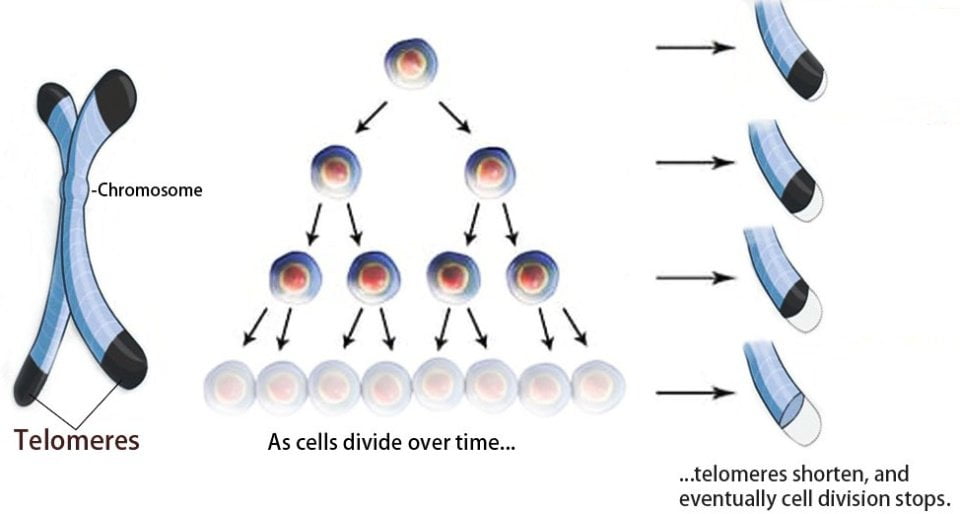
Telomeres, protein-DNA complexes localized at the ends of linear chromosomes, are the primary gatekeepers of the genome stability. Their length shortens with each cell division and correlates inversely with increasing age. The length can be modified by genetic and epigenetic factors, sex hormones and inflammatory reactions. Since the telomeres are G-C rich tandem repeats, they are also sensitive to oxidative stress. A critical minimum telomere length triggers a cell cycle arrest or cell senescence[1]. Therefore, telomere length can serve as a biological indicator and a determining factor of the replicative capacity of immune cells[2].
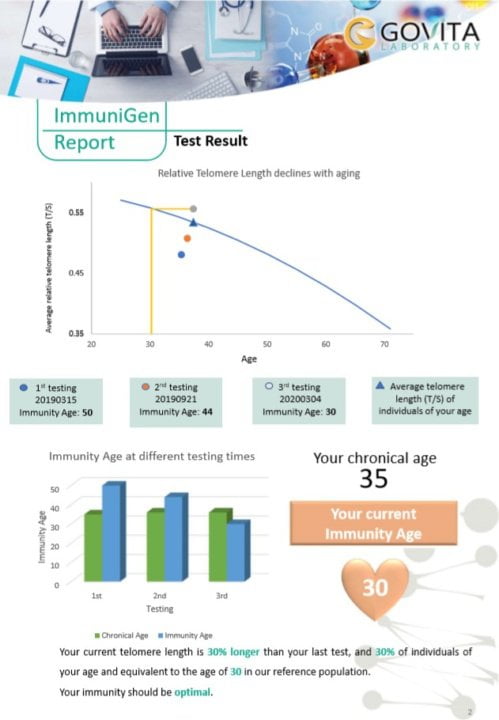
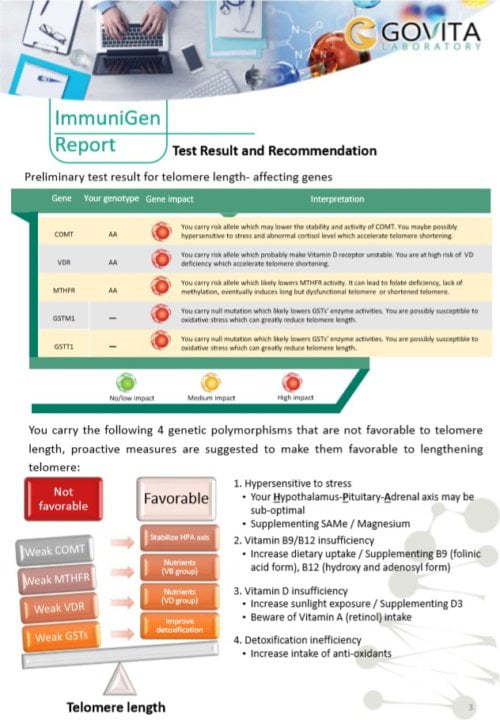
Our ImmuniGen test is performed from a fingertip dried blood spot of easily and pain-less sampling. It measures the telomere length of peripheral blood mononuclear cells including immune cells in dry blood spots by qPCR. The method provides a relative telomere length (T) compared to a single (T) gene and the results are expressed as T/S ratio. T/S is then matched against our database with a large reference population and compared to that of individuals having the same chronological age to the tested person to monitor telomere shortening. In addition, we test the genetic make-up which affects the telomere length and provide corresponding solutions to make it favour the telomere lengthening.
Telomere Shortening Factors


[1] Kaszubowska, L. Telomere shortening and ageing of the immune system. J. Physiol. Pharmacol. 59, 169–186 (2008).
[2] Allsopp RC, et al. Telomere length predicts replicative capacity of human fibroblasts. Proc Natl Acad Sci USA. 89: 10114-10118 (1992).
[3] Ornish D, et al. Effect of comprehensive lifestyle changes on telomerase activity and telomere length in men with biopsy-proven low-risk prostate cancer: 5-year follow-up of a descriptive pilot study. Lancet Oncol. 14:1112-1120 (2013).
[3] Jiang, Y. et al. Basal cortisol, cortisol reactivity, and telomere length: A systematic review and meta-analysis. Psychoneuroendocrinology 103, 163–172 (2019).
[4] Walder, D. et al. Catechol-O-Methyltransferase Modulation Of Cortisol Secretion In Psychiatrically At-Risk And Healthy Adolescents. Psychiatr Genet 20, 166–170 (2010).
[5] Pusceddu, I. et al. The role of telomeres and vitamin D in cellular aging and age-related diseases. Clin Chem Lab Med 53, 1661-78 (2015).
[6] Bull, C. F. et al. Folate deficiency induces dysfunctional long and short telomeres; Both states are associated with hypomethylation and DNA damage in human WIL2-NS cells. Cancer Prev Res 7, 128–138 (2014).
[7] Shen, J. et al. Telomere length, oxidative damage, antioxidants and breast cancer risk. Atherosclerosis 124, 1637–1643 (2010).
[8] Strandberg, T. E. et al. Association between alcohol consumption in healthy midlife and telomere length in older men. The Helsinki Businessmen Study. Eur. J. Epidemiol. 27, 815–822 (2012).
[9] Valdes, A. M. et al. Obesity, cigarette smoking, and telomere length in women. Lancet 366, 662–664 (2005).
[10] Cassidy, A. et al. Associations between diet, lifestyle factors, and telomere length in women. Am. J. Clin. Nutr. 91, 1273–1280 (2010).
[11] Tucker, L. A. Dietary fiber and telomere length in 5674 U.S. adults: An NHANES study of biological aging. Nutrients 10, 1–16 (2018).
[12] Starkweather, A.R., et al. An Integrative Review of Factors Associated with Telomere Length and Implications for Biobehavioral Research. Nurs Res 63, 36-50 (2014).
[13] Shammas, M. Telomeres, lifestyle, cancer, and aging. Curr Opin Clin Nutr Metab Care 14, 28-34 (2011).
[14] Chen, Y. et al. Short leukocyte telomere length predicts poor prognosis and indicates altered immune functions in colorectal cancer patients. Ann. Oncol. 25, 869–876 (2014).
[15] Najarro, K. et al. Telomere length as an indicator of the robustness of B- and T-cell response to influenza in older adults. J. Infect. Dis. 212, 1261–1269 (2015).
[16] Helby, J., et al. Shorter leukocyte telomere length is associated with higher risk of infections: A prospective study of 75,309 individuals from the general population. Haematologica 102, 1457–1465 (2017).
[17] Wu, X. et al. Telomere dysfunction: a potential cancer predisposition factor. J. Natl. Cancer Inst. 95, 1211–8 (2003).
[18] Haycock, P. C. et al. Leucocyte telomere length and risk of cardiovascular disease: Systematic review and meta- Analysis. BMJ 349, 1–11 (2014).
[19] Ding, H. et al. Telomere length and risk of stroke in Chinese. Stroke 43, 658–63 (2012).
[20] Zhang, W. et al. Short telomere length in blood leucocytes contributes to the presence of atherothrombotic stroke and haemorrhagic stroke and risk of post-stroke death. Clin Sci 125, 27–36 (2013).
[21] Jiang, X. et al. Decreased leukocyte telomere length (LTL) is associated with stroke but unlikely to be causative. PLoS ONE. 8, e68254 (2013).
Govita fights COVID-19 pandemic together with you!